@GroupParrinello IIT ETHZ | PhD @QUBelfast.
Computational physicist, #MolecularDynamics #Biophysics

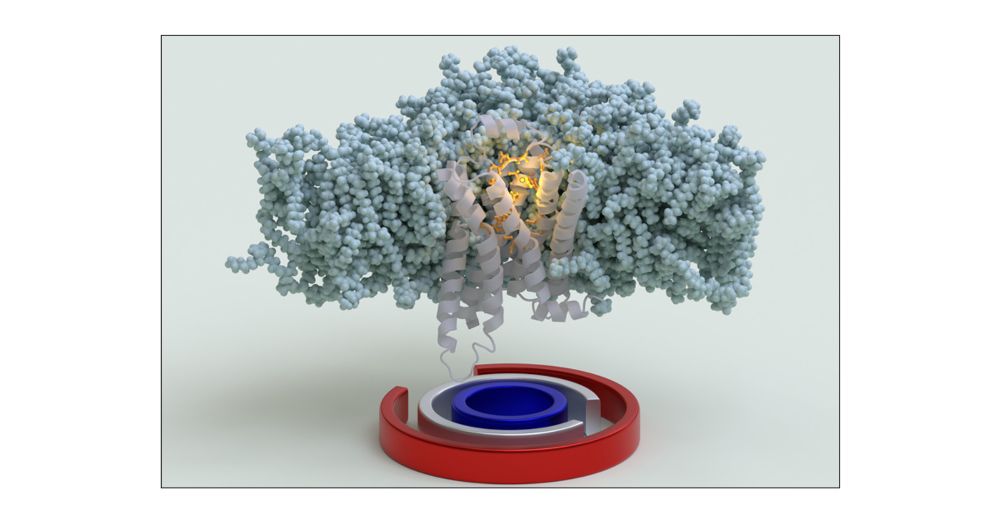
With @saureli.bsky.social @valeriorizzi.bsky.social and Nicola we used OneOPES to fully converge the free energy landscapes associated with B1AR apo/holo activation.
With @saureli.bsky.social @valeriorizzi.bsky.social and Nicola we used OneOPES to fully converge the free energy landscapes associated with B1AR apo/holo activation.

