PhD|Institute Jacques Monod, Paris. Passionate about ECM mechanics, Intermediate filaments, and cell migration.
bit.ly/4oS2Ju3


www.biorxiv.org/content/10.1...
See the thread 🧵 below for more information.

www.biorxiv.org/content/10.1...
See the thread 🧵 below for more information.

www.rockefeller.edu/news/38446-h...

www.rockefeller.edu/news/38446-h...
1/
doi.org/10.1101/2025...
1/
doi.org/10.1101/2025...
Gregor & co use tunable hydrogels to show cell motility, not gene expression, drives axis formation in gastruloids
journals.biologists.com/dev/article/...
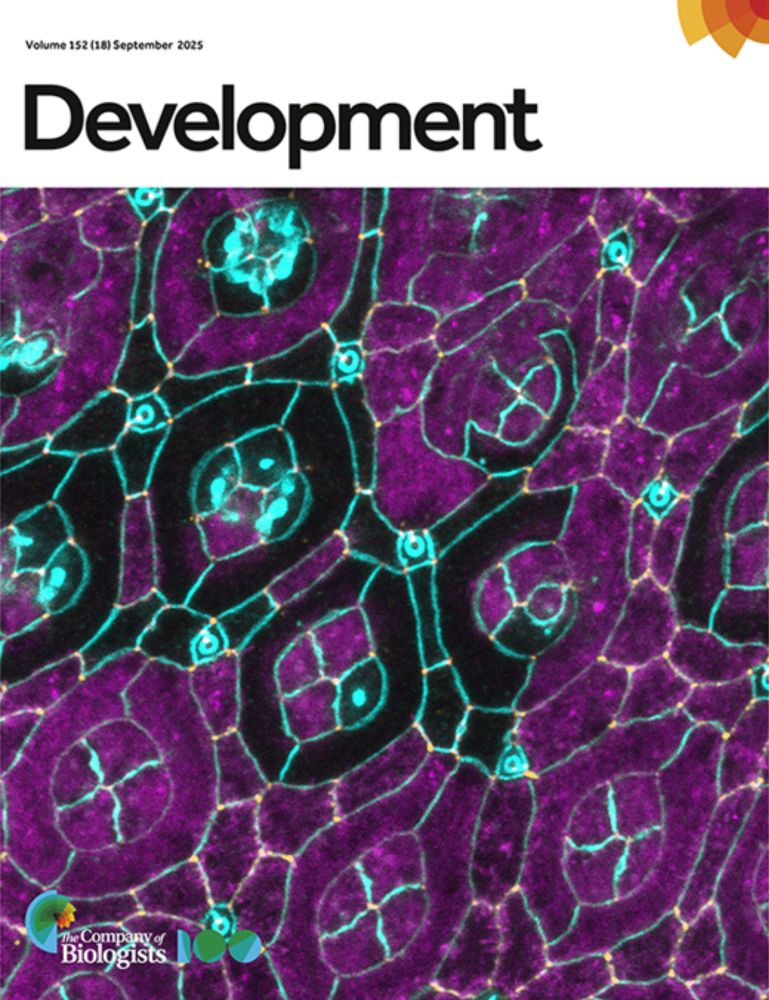
Gregor & co use tunable hydrogels to show cell motility, not gene expression, drives axis formation in gastruloids
journals.biologists.com/dev/article/...
Special thanks to the reviewers whose comments improved our manuscript a lot! rdcu.be/eI3tD

Special thanks to the reviewers whose comments improved our manuscript a lot! rdcu.be/eI3tD
@sreejithsanthosh.bsky.social
biorxiv.org/content/10.1...
@sreejithsanthosh.bsky.social
biorxiv.org/content/10.1...
Congrats to Huiqiong Wu, G. Arkowitz and R. Chilupuri!
www.biorxiv.org/content/10.1...

Congrats to Huiqiong Wu, G. Arkowitz and R. Chilupuri!
www.biorxiv.org/content/10.1...


We combined microfluidics, proteomics, and RNA-seq to map spatiotemporal protein expression during mouse somitogenesis and found a novel regulatory strategy: dynamic antagonistic gradients fine-tune signalling strength.
doi.org/10.1101/2025...

We combined microfluidics, proteomics, and RNA-seq to map spatiotemporal protein expression during mouse somitogenesis and found a novel regulatory strategy: dynamic antagonistic gradients fine-tune signalling strength.
doi.org/10.1101/2025...

www.biorxiv.org/content/10.1...

www.biorxiv.org/content/10.1...




www.biorxiv.org/cgi/content/...
www.biorxiv.org/cgi/content/...
For more information, please contact: marie-helene.verlhac@college-de-france.fr

For more information, please contact: marie-helene.verlhac@college-de-france.fr
🔗 www.nature.com/articles/s41...
#biophysics #syntheticcell #softmatter
🔗 www.nature.com/articles/s41...
#biophysics #syntheticcell #softmatter

Super fun collab with @davidbrueckner.bsky.social and @gcharras.bsky.social 👏🏻
Enjoy the reading www.nature.com/articles/s41...

Super fun collab with @davidbrueckner.bsky.social and @gcharras.bsky.social 👏🏻
Enjoy the reading www.nature.com/articles/s41...

