
https://sites.google.com/ualberta.ca/theditranigroup/home
https://scholar.google.ca/citations?user=LeG-9koAAAAJ&hl=en
www.nature.com/articles/s41...

www.nature.com/articles/s41...
pubs.acs.org/doi/full/10....

pubs.acs.org/doi/full/10....
U Alberta is looking for a director for their new #cryoEM facility
Support excellent researchers (who are really nice people) using state-of-the-art infrastructure.
+ reasonable cost of living
+ near some of most beautiful places in world
+ 🇨🇦
apps.ualberta.ca/careers/post...

U Alberta is looking for a director for their new #cryoEM facility
Support excellent researchers (who are really nice people) using state-of-the-art infrastructure.
+ reasonable cost of living
+ near some of most beautiful places in world
+ 🇨🇦
apps.ualberta.ca/careers/post...
@hanlinw222.bsky.social's 1st struct of RAVE bound to a partial V1 complex now out in PNAS.
www.pnas.org/doi/10.1073/...
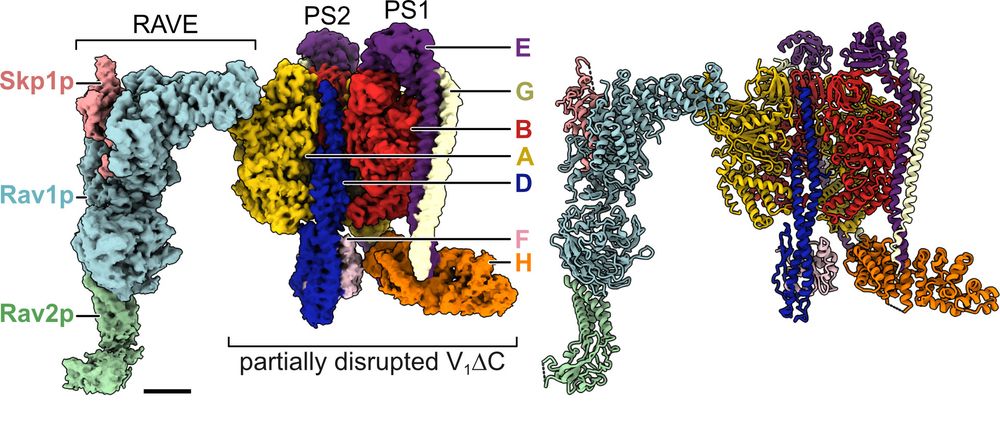
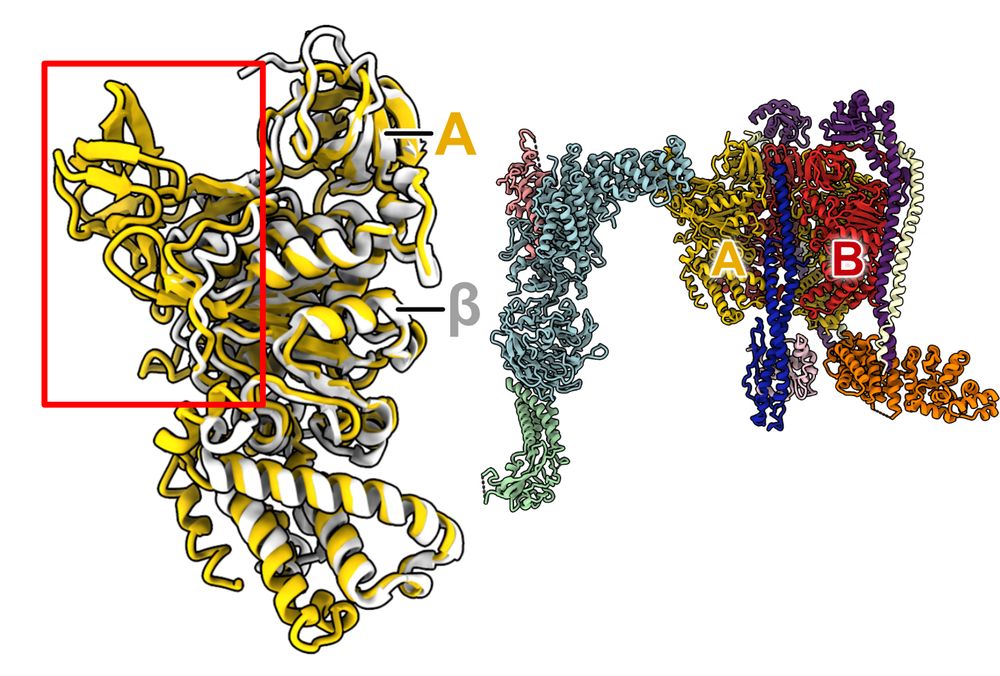
@hanlinw222.bsky.social's 1st struct of RAVE bound to a partial V1 complex now out in PNAS.
www.pnas.org/doi/10.1073/...

